(Associated Press)
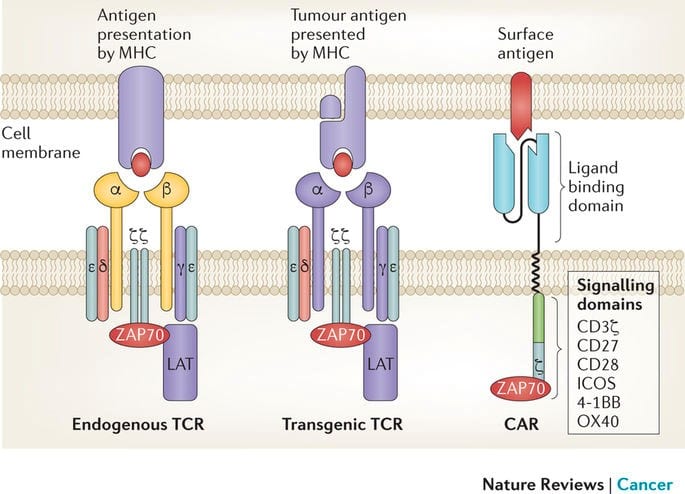
T cells recognize and kill their targets, usually pathogens, through their T cell receptor. When the T cell receptor recognizes a specific molecule on a bacteria or virus, it signals to the T cell to kill the invader. Additional receptors, when stimulated, tell the T cell to proliferate or perform other functions. Chimeric antigen receptors (CARs) are created using the principle signaling components of the T cell receptor and costimulation molecules. Rather than recognizing a pathogen, the CAR is engineered to recognize a tumor antigen instead, and the internal domain signals the T cell to kill the target cell and proliferate until no more targets remain.
First generation CARs consisted of a TCR stimulatory domain attached to a single chain variable antibody fragment (1). The subsequent addition of a costimulatory domain (second generation CAR) has greatly enhanced its success by improving T-cell survival and proliferation when it encounters antigen-expressing target cells (2). The use of CAR-modified T cells that target CD19, a B cell receptor, has been shown to induce durable remissions in patients with chemorefractory B cell malignancies (3-8).
Featured Image (top): A CAR T cell (right) recognizes a tumor cell (left).
References
- Gross G, et al. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci U S A. 1989.
- Maher J, et al. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotechnol. 2002.
- Davila ML, et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Science translational medicine. 2014.
- Kochenderfer JN, et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J Clin Oncol. 2015.
- Grupp SA, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013.
- Porter DL, et al. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med. 2011.
- Maude SL, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014.
- Park JH, et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N Engl J Med. 2017.
- Fesnak AD, et al. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer. 2016.